Ice soften salt does extra for winter security than most individuals suppose. Winter can look calm and exquisite, however it could possibly additionally flip roads and walkways into slick, harmful surfaces quick. A sudden chilly entrance could make streets really feel like glass and sidewalks really feel unsafe.
Snowplows and grit vehicles play their half, however ice soften salt does the quiet, regular work. This small mineral helps break the bond between ice and the bottom, making it simpler for folks and automobiles to maneuver safely.
From small driveways to huge highways, ice soften salt is used all over the place. However there may be extra to it than a number of grains scattered on the bottom. Behind every crystal is a mixture of chemistry, temperature, and security wants. Ice soften salt isn’t just a easy product. It’s a instrument formed by science and years of testing to maintain winter journey safer for everybody.
That is the science that retains cities transferring when the world freezes.
What Ice Soften Salt Truly Does: Breaking the Winter “Ice Lock”
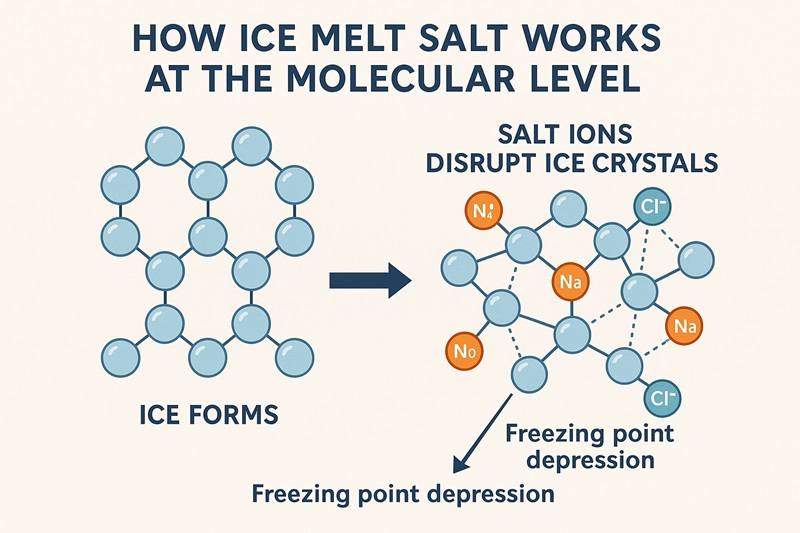
To grasp ice soften salt, you will need to begin with water—essentially the most extraordinary, but most extraordinary chemical on Earth. Pure water freezes at 32°F (0°C), however winter not often presents pure water or pure circumstances. As quickly as salt begins to dissolve into melting snow or frost, it triggers a course of often called freezing level melancholy.
This phenomenon is fantastically easy:
- Water molecules have to align right into a crystal lattice to turn out to be ice.
- Salt dissolves into charged particles (ions).
- These ions disrupt the formation of that lattice.
- The water stays liquid at temperatures the place it will usually freeze.
On a molecular stage, it’s as if the salt scatters tiny obstacles throughout the trail of forming ice crystals—breaking the construction earlier than it could possibly harden.
Some salts go additional, releasing warmth as they dissolve. These “exothermic” de-icers begin melting on contact, even in dangerously low temperatures. Others are “endothermic,” absorbing warmth as a substitute—efficient solely when a faint hint of environmental heat is out there.
This distinction between releasing warmth and absorbing it explains why some salts soften ice immediately whereas others flip sluggish when the chilly turns into excessive.
Exothermic vs. Endothermic: Why Some Salts Appear to “Burn Via” Ice
Not all ice soften merchandise are created equal. Their chemical habits adjustments dramatically as temperatures dip.
Endothermic Melts: Sluggish, Dependable, Temperamental
Sodium chloride, or conventional rock salt, dominates international winter upkeep. It’s plentiful, low-cost, and enough for common chilly. Nevertheless it’s endothermic—it attracts warmth from its atmosphere—and when temperatures fall under 10–15°F, it begins to lose its capability to activate.
Exothermic Melts: The Powerhouses of Subzero Chilly
Chemical substances like calcium chloride and magnesium chloride dissolve with a launch of warmth—typically sufficient to really feel heat in your gloves.
Their benefits are hanging:
- On the spot brine formation
- Efficient far under zero
- Quick melting motion
- Dependable underneath harsh freeze-thaw cycles
In deep winter, these salts don’t merely soften ice—they chemically overpower it.
The Most Frequent Ice Soften Salts—and the Temperatures They Dominate
Winter roads depend on a handful of salts, every with its personal candy spot. Consider them as winter instruments, every one shining at a distinct temperature vary.
Sodium chloride—higher often called rock salt—is the traditional alternative. Most individuals use it as a result of it’s low-cost and simple to seek out, and it really works properly so long as temperatures keep above about 20°F (–6°C). As soon as the chilly turns into extra intense, although, it slows down and loses a lot of its energy.
When the climate dips far under zero, calcium chloride steps in. It’s the heavy-hitter of the group, kicking into gear even at –25°F (–32°C). This salt releases warmth when it touches moisture, which is why it appears to “jump-start” the melting course of. The trade-off? It may be harsh on pores and skin and will go away its mark on sure surfaces.
Magnesium chloride tends to be the selection for houses with pets, gardens, or numerous concrete. It really works all the way down to –13°F (–25°C), and though it’s not the quickest melter, it’s kinder to surrounding vegetation and fewer corrosive than the harsher choices.
Then there’s potassium chloride, which performs finest when used as a part of a mix. By itself, it stops being efficient as soon as the temperature falls close to 20°F (–6°C), so folks typically use it when they need a milder, lower-impact remedy relatively than a deep-freeze resolution.
Lastly, premium blends mix a number of salts to steadiness velocity, security, and cold-weather efficiency. The precise temperature vary depends upon the components, however many householders like them as a result of they scale back floor harm and soften ice quicker than a single salt alone.
Ice Soften Salt Comparability
| Ice Soften Sort | How Nicely It Handles the Chilly | The place It Works Finest | Actual-World Benefits | Sensible Drawbacks |
| Sodium Chloride (Rock Salt) | Works reliably till temps fall close to 20°F (–6°C) | Driveways, metropolis streets, on a regular basis winter circumstances | Price range-friendly, simple to seek out in bulk | Slows down rather a lot throughout deep chilly; not ultimate for harsh freezes |
| Calcium Chloride (CaCl₂) | Stays efficient even at –25°F (–32°C) | Excessive-cold areas, late-night freeze occasions | “Warmth burst” response begins melting quick; nice for emergencies | Can irritate pores and skin and could also be tough on sure surfaces |
| Magnesium Chloride (MgCl₂) | Performs all the way down to about –13°F (–25°C) | Properties with pets, gardens, or delicate concrete | Gentler on vegetation, much less corrosive than harsher salts | Melts ice extra slowly than CaCl₂; reasonable efficiency |
| Potassium Chloride (KCl) | Stops working close to 20°F (–6°C) | Gentle winters, blended merchandise | Softer, lower-impact possibility when excessive chilly isn’t a priority | Poor low-temperature efficiency by itself |
| Premium Blends | Relies on the combo; usually covers a broad temperature span | Householders wanting effectivity + floor safety | Balanced melting velocity, diminished floor harm, stable all-around alternative | Worth varies; high quality depends upon model and mix ratio |
Why Temperature Guidelines All the pieces
Ice soften salt doesn’t function in a vacuum. It responds to temperature, moisture, daylight, and stress.
Temperature: The Gatekeeper of Melting
Rock salt slows dramatically because the air drops under 10°F.
Calcium chloride thrives the place others fail.
Moisture Ranges: The Hidden Variable
Salt solely works when it could possibly turn out to be brine—a salty liquid that breaks ice’s bond.
- Dry ice: sluggish activation
- Moist ice: instantaneous melting
Daylight & Darkish Surfaces
Even a number of levels of radiant warmth could make the distinction between failure and success.
Site visitors & Foot Stress
Each automobile tire and boot heel presses salt deeper into ice, accelerating melting.
Collectively, these forces flip a static winter atmosphere right into a micro-laboratory of chemical change.
Tips on how to Use Ice Soften Salt Appropriately
Salt doesn’t soften quicker simply since you throw extra on the floor. In truth, over-salting is among the commonest—and expensive—errors folks make.
Appropriate Utility Charge
Specialists suggest:
- Householders: ½ cup per sq. yard
- Parking heaps / heavy ice: Modify primarily based on thickness
Extra salt doesn’t create “extra melting” as soon as the brine saturates.
Pre-Therapy: The Secret of Municipal Success
Cities throughout North America now pre-treat roads earlier than storms. This creates a skinny brine barrier on the pavement:
- Ice can’t kind a good bond
- Roads stay plowable
- Black ice is much less possible
- Complete salt use drops dramatically
This methodology is each safer and extra economical.
Environmental and Materials Harm: The Worth We Pay for Security
Salt is efficient—however not innocent.
Soil & Water
Salt melts ice properly, but it surely leaves many issues behind. In soil and water, chloride doesn’t wash away quick. It builds up over time. This additional salt adjustments how soil holds water and vitamins. Crops begin to really feel burdened. Their roots weaken. Some vegetation cease rising in any respect.
Runoff doesn’t keep the place it begins. Melted salt water strikes by drains and ditches into ponds, streams, and even groundwater. As chloride ranges rise, fish, bugs, and different small life types battle to outlive. Entire water methods can fall out of steadiness.
Concrete Harm
Salt additionally harms our roads and sidewalks. When salty water slips into tiny concrete pores and freezes, it expands. This stress pushes the concrete aside. One freeze does little, however many freeze–thaw cycles trigger cracks and crumbling edges.
Corrosion
Metallic takes successful too. Salt makes rust kind quicker. Vehicles, handrails, instruments, bridges, and different metal constructions break down extra rapidly in winter. The restore prices add up quick, from small fixes at house to huge metropolis initiatives.
Salt retains us protected on winter roads, however the harm it leaves behind stays lengthy after the snow melts.
When Ice Soften Stops Working
Beneath sure thresholds, even the strongest salts face bodily limits.
Molecular Motion Slows
In excessive chilly, water molecules transfer too slowly for some salts to dissolve.
Brine Re-Freezes
Each salt has a eutectic level—its lowest doable melting temperature. Beneath this, the brine itself begins to freeze.
Mechanical Elimination Turns into Necessary
Beneath –25°F (–32°C):
- Shoveling
- Scraping
- Sanding for traction
- Heavy plowing
…turn out to be the first types of ice management.
Even chemistry has limits.
Ice Soften Salt on Roads: The Spine of Winter Transportation
Salt is the quiet drive that retains winter roads open. On busy streets and highways, ice soften salt works within the background so automobiles and vehicles can hold transferring. It could look easy, however it’s nonetheless top-of-the-line instruments for retaining roads protected when the temperature drops.
How Salt Helps Ice Raise Off the Highway
Salt will get to work as quickly because it touches a little bit of moisture. It turns into a skinny brine. This brine slides between the ice and the highway. As soon as it does, the ice loses its grip. New ice additionally has a tougher time sticking. Snowplows can then push the snow and ice away as a substitute of combating a tough, frozen layer caught to the pavement.
The Varieties of Ice Soften Salt Cities Use
Highway crews use completely different salts primarily based on the climate.
Rock salt is the most typical as a result of it’s low-cost and dependable.
When it will get colder, crews swap to calcium chloride as a result of it offers off warmth and works quicker.
Magnesium chloride is utilized in busy areas the place folks fear about soil and plant well being, since it’s simpler on the atmosphere.
Many cities additionally spray liquid brine earlier than a storm to cease ice from forming within the first place.
What Occurs When It Will get Too Chilly
Salt can’t work properly in excessive chilly. As soon as the air falls under –20°F, melting slows rather a lot. When that occurs, crews change their plan. They scrape the roads extra typically. They unfold sand to assist automobiles get traction. Additionally they use liquid anti-icers to sluggish the formation of black ice. Plows run extra typically, counting on drive as a substitute of salt till the climate warms up.
Selecting the Proper Ice Soften Salt
Finest for Excessive Chilly:
Calcium chloride
Quickest soften, most dependable under 0°F.
Finest for Driveways and Sidewalks:
Magnesium chloride
Gentler on concrete, vegetation, and pets.
Finest Price range Choice:
Rock salt (sodium chloride)
Reasonably priced, efficient above ~15°F.
Finest Eco-Pleasant Choice:
CMA (Calcium Magnesium Acetate) or blended “inexperienced” melts
Low corrosion, protected for vegetation.
Finest Pet-Secure Choice:
Magnesium chloride or natural blends
Decrease burn threat, smoother granules.
Frequent Errors Folks Make With Ice Soften
Even well-intentioned householders misuse salt.
Listed below are essentially the most frequent errors:
- Making use of salt earlier than clearing snow
- Dumping extreme salt on one spot
- Utilizing rock salt in excessive chilly
- Mixing incompatible chemical compounds
- Failing to retailer salt hermetic
- Utilizing salt on newly poured concrete
- Forgetting to reapply after heavy site visitors
Avoiding these errors will increase security whereas decreasing harm and value.
Pet Security: Defending Paws in Winter
Some salts irritate paws resulting from alkaline pH or sharp granules.
To guard animals:
- Select magnesium chloride blends
- Use canine booties
- Wash paws after walks
- Lay down protecting mats close to entrances
Pets depend on human consideration to remain protected in winter environments.
Tips on how to Retailer Ice Soften Correctly
Salt is hygroscopic—it absorbs moisture from the air.
Retailer it in:
- Hermetic containers
- Cool, dry areas
- Elevated surfaces away from concrete flooring
Clumped salt loses effectiveness and spreads inconsistently.
FAQ
Does ice soften salt work under zero?
Sure—calcium chloride and magnesium chloride stay efficient in subzero temperatures.
Is ice soften unhealthy for concrete?
Chloride salts speed up freeze–thaw harm over time.
What’s the most secure ice soften for pets?
Magnesium chloride and natural pet-safe blends.
How lengthy does ice soften take to work?
Wherever from 5–20 minutes relying on temperature, salt sort, and ice thickness.
Is rock salt the identical as ice soften?
Rock salt is one sort. Blends carry out higher in excessive chilly and supply improved security.
Conclusion: The Invisible Chemistry That Makes Winter Livable
Winter feels safer due to a easy little bit of chemistry. Each clear sidewalk and open highway depends upon how salt reacts with ice and chilly. Ice soften salt could look plain, but it surely performs an enormous function in retaining day by day life transferring.
Once we understand how every sort of salt works—and when it stops working—we are able to use it in smarter, safer methods. With the appropriate decisions, winter turns into simpler to deal with. It exhibits how tiny salt crystals could make an enormous distinction, serving to cities hold going even when the world freezes.
Meta: