OGG synthesis was achieved by GG oxidation utilizing sodium periodate (NaIO4). Structural affirmation was obtained by 1H nuclear magnetic resonance (1H NMR) and Fourier rework infrared spectroscopy (FTIR) analyses. The ¹H NMR spectrum revealed a attribute aldehydic proton peak at 9.3 ppm (Determine S1(a)). The FTIR spectrum of pure GG (Determine S1(b)) revealed typical peaks at 3400 (O–H), 2910 (C–H), 1152 (C-O), and 1002 cm⁻¹ (glycosidic linkage of the GG pyranose ring). After the response of GG with NaIO4, a brand new peak was noticed at 1727 cm–1, similar to the aldehyde symmetrical vibration. The aldehyde teams of the monomeric sugar items and the OH teams of the unoxidized ring residue can kind a hemi acetal bond through the chemical response, which seemingly accounts for the comparatively weak depth of aldehyde group alerts. Equally, CMCS synthesis was verified by comparative 1HNMR and FTIR analyses of CS and CMCS. The 1HNMR spectrum confirmed attribute peaks at 3.2–3.5 ppm for H-2 protons of carboxymethyl teams (Determine S1(c)). FTIR evaluation revealed vital modifications within the chemical construction, the place the attribute amino group bending vibration at 1550 cm⁻¹ and C-O-C uneven stretching at 1242 cm⁻¹ in pristine CS have been accompanied by a brand new absorption band at 1422 cm⁻¹ in CMCS, similar to the uneven stretching vibration of carboxylate teams (Determine S1(d)). These spectral options collectively display the profitable introduction of carboxymethyl teams onto the chitosan spine, confirming the synthesis of CMCS.
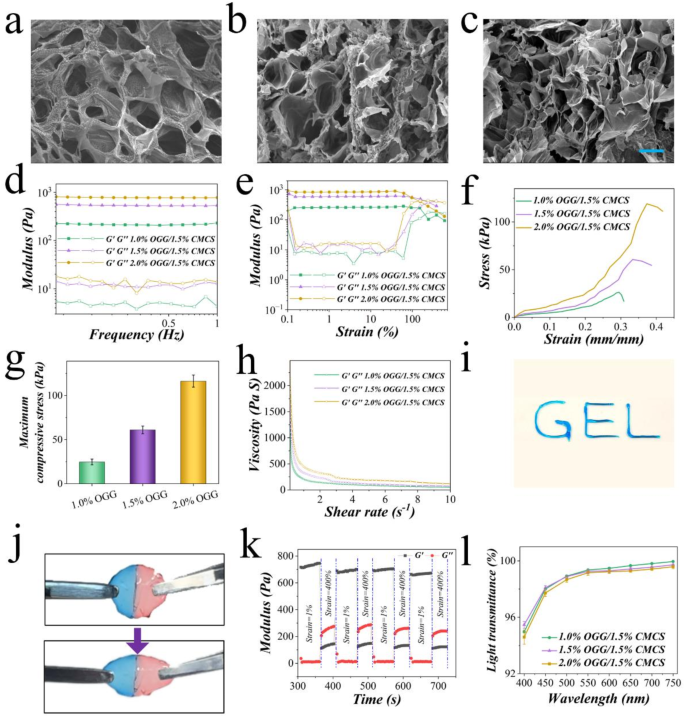
The fabrication of OGG/CMCS hydrogel concerned easy mixing of OGG and CMCS options because the aldehyde group of OGG can kind reversible Schiff base bonds with the amino group of CMCS. Hydrogels ready by 1.5 wt% CMSC and ranging concentrations of OGG (2.0 wt%, 1.5 wt%, and 1.0 wt%) have been used to carry out the in vitro physiochemical properties characterization. The microstructure of the hydrogels was characterised by observing freeze-dried samples utilizing scanning electron microscopy (Figs. 2(a)–2(c)). Variations within the formulation of the hydrogel have been discovered to influence its porosity. Particularly, the OGG/CMCS-2.0% hydrogel demonstrated smaller pore sizes and a denser community construction compared to the OGG/CMCS-1.5% and OGG/CMCS-1.0% (Determine S2).
Moreover, good homogeneity of the hydrogel performs an important position in enhancing its mechanical power, whereas additionally offering a extra appropriate surroundings for cell adhesion and proliferation [18]. Consequently, dynamic rheology evaluation was performed on OGG/CMCS hydrogels with totally different formulations to additional examine these properties. For every hydrogel, its storage modulus (G’) exceeded the loss modulus (G”) persistently, signifying that the hydrogel was free-standing (Fig. 2(d)). The modulus–pressure curves demonstrated that beneath strains lower than 100%, the modulus of all hydrogels remained regular, with G’ surpassing G”, suggesting the community construction was secure throughout the vary (Fig. 2(e)). As pressure elevated, the G’ declined, finally falling under G”, indicating the breakdown of the community construction. In addition to, the OGG/CMCS-2.0% demonstrated the best yielding modulus, whereas OGG/CMCS-1.0% exhibited the bottom yielding modulus, indicating a correlation between the mechanical properties and microscopic constructions of the hydrogel. The compressive mechanical properties of the hydrogels have been quantitatively evaluated, as illustrated in Figs. 2(f) and a couple of(g). A concentration-dependent enhancement in compressive power was noticed, with OGG/CMCS-2.0% exhibiting the best most compressive power, adopted by OGG/CMCS-1.5%, whereas OGG/CMCS-1.0% demonstrated the bottom mechanical power. This pattern aligns with the rheological measurements.
Self-healing injectable hydrogels have intensive functions, together with drug supply and tissue regeneration [19], and supply an answer for the restore of an irregular-shaped corneal wound. Consequently, we assessed the rheological characterization of the OGG/CMCS hydrogels. All formulations exhibited pronounced shear-thinning habits, with viscosity reducing as shear charges elevated beneath a relentless 1% pressure (Fig. 2(h)), indicating glorious injectability. This rheological property allows {smooth} extrusion by 25-gauge needles whereas sustaining structural integrity, as demonstrated in Fig. 2(i), with out clogging. To substantiate the self-healing functionality of OGG/CMCS hydrogels, we fabricated two shell-shaped hydrogels containing pink and blue dyes (Determine S3). The hydrogels have been reduce with a blade into two half items, and one half containing pink dye was carefully introduced into contact with the one containing blue dye. After 2 hours, the 2 halves seamlessly healed right into a unified construction, able to supporting its weight beneath gravity (Determine S3). The healed unit additionally demonstrated the power to resist stretching as much as nearly twice its unique size with out rupturing (Fig. 2(j)). Furthermore, steady oscillatory step-strain measurements have been performed to check the rheological self-healing properties. Throughout the take a look at, the oscillatory pressure transitioned from 1% over an 80-second interval to 400% over a 60-second interval, with a complete of 4 cycles. When the pressure reached 400%, G’ grew to become lower than G”, indicating hydrogel breakage. But, G’ promptly returned to the preliminary worth because the pressure reverted to 1%, surpassing G” (Fig. 2(ok)). The noticed self-healing functionality facilitates each {smooth} injection by narrow-gauge needles and efficient integration with corneal defects, enabling minimally invasive restore.
Fabrication and characterization of the OGG/CMCS hydrogels. (a)–(c) Scanning electron microscopy pictures depicting OGG/CMCS hydrogels with OGG concentrations of 1.0% (a), 1.5% (b), and a couple of.0% (c), respectively. Scale bar: 20 µm. (d)-(e) Frequency-dependent (d) and strain-dependent (e) rheology evaluation of the hydrogels. (f) Compressive stress-strain profiles of the hydrogels. (g) Histogram of the utmost compressive stress of hydrogels. (h) Viscosity adjustments in hydrogels with rising shear charges. (i) Injectability and extrudability of the hydrogel by a syringe. (j) Stretchability evaluation of the hydrogel. (ok) Variation of G’ and G’’ with time throughout steady step pressure measurement in 1% and 400% pressure cycles. (l) Mild transmittance of the hydrogels throughout wavelengths from 400 to 750 nm.
Notably, reaching excessive transparency in seen mild is essential for a corneal patch to keep away from hindering imaginative and prescient throughout corneal restore. Thus, the sunshine transmittance of OGG/CMCS hydrogels was evaluated on totally different wavelengths (Fig. 2(l)). All hydrogel samples with totally different formulations exhibited outstanding transparency, though the transmittance skilled a slight lower with the rise in OGG focus. The swelling and degradation traits of OGG/CMCS hydrogels have been additionally evaluated. The OGG/CMCS-2% formulation demonstrated decrease swelling capability (Determine S4(a)) and slower degradation (Determine S4(b)) throughout incubation in PBS in comparison with each OGG/CMCS-1.5% and OGG/CMCS-1% hydrogels, suggesting a concentration-dependent habits.
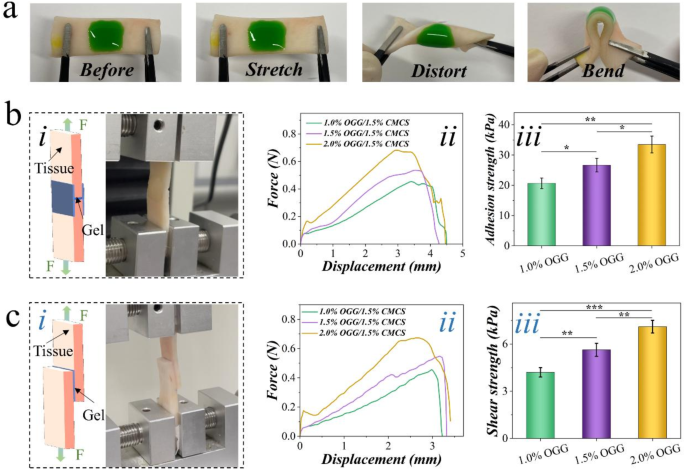
Moreover, the OGG/CMCS hydrogels demonstrated efficient tissue adhesive properties. The green-dye-containing hydrogels have been in situ shaped on porcine pores and skin, displaying strong adherence beneath stretch, distortion, and bending (Fig. 3(a)). After immersing in water, the hydrogels might nonetheless adhere to porcine pores and skin, even beneath distortion (Determine S5). This implies a sturdy tissue-binding capability of the hydrogel. Additional comparisons of adhesiveness amongst totally different formulations have been performed by wound closure and lap shear exams. The OGG/CMCS-2% hydrogel demonstrated considerably increased adhesion power (33.5 ± 2.78 kPa) than each OGG/CMCS-1.5% (26.7 ± 2.25 kPa, p < 0.05) and OGG/CMCS-1% hydrogels (20.7 ± 1.76 kPa, p < 0.01) (Fig. 3(b)). Equally, OGG/CMCS-2% hydrogel demonstrated considerably increased shear power (7.1 ± 0.4 kPa) than each OGG/CMCS-1.5% hydrogel (5.6 ± 0.4 kPa, p < 0.01) and OGG/CMCS-1% hydrogel (4.2 ± 0.3 kPa, p < 0.001) (Fig. 3(c)), suggesting the adhesive properties exhibited a concentration-dependent habits.
Adhesion properties. (a) Images of OGG/CMCS hydrogels adhered to porcine pores and skin tissue. The hydrogel stays intact with out detachment or cracking with the tissue, even throughout stretching, distorting, or bending. (b)-(c) Tissue adhesive properties of the hydrogels: (i) schematic illustration and images of the take a look at; (ii) force-displacement curves; (iii) histogram of the adhesion (b) and shear power (c) (*p < 0.05, **p < 0.01, ***p < 0.001)
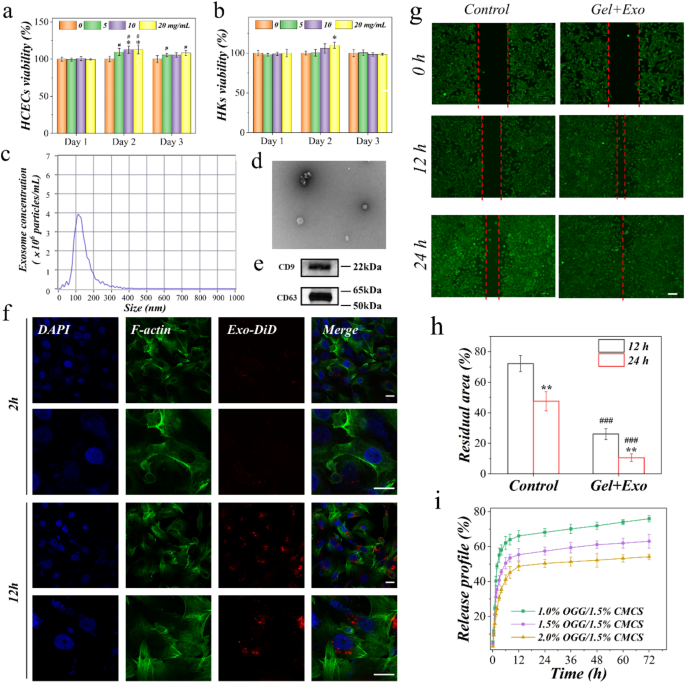
To evaluate biocompatibility, the liquid extracted from the OGG/CMCS hydrogel was utilized for culturing human corneal epithelial cells (HCECs) and keratocytes (HKs). Cells have been cultured with hydrogel extracts (0, 5, 10, and 20 mg/mL) individually and cell viability was evaluated on days 1, 2, and three utilizing the CCK-8 assay. On day 2, the viability of HCECs in extract liquid in any respect three concentrations was considerably increased than the management (regular medium), and the viability of HKs in extract liquid at 20 mg/mL was considerably increased than the management (Figs. 4(a) and 4(b), all p < 0.05). The viability remained excessive for 3 days, indicating the hydrogel’s strong biocompatibility. In comparison with different self-healing hydrogels and associated hydrogel programs for corneal regeneration, our work displays comparatively robust mechanical power, strong adhesion, and glorious cytocompatibility [20,21,22,23,24] (Determine S6).
To guage the standard of MSC-Exos, their focus was quantified utilizing nanoparticle monitoring evaluation (NTA), revealing an unique focus of seven.3 × 1010 particles/mL (Fig. 4(c)). Moreover, the morphology of MSC-Exos was noticed through transmission electron microscopy (TEM). The acquired Exos exhibited a attribute saucer form, that includes small vesicles with a central despair, according to prior observations (Fig. 4(d)). Moreover, the MSC-Exos expressed CD9 and CD63 exosomal markers (Fig. 4(e)). Following a 12-hour co-culture of MSC-Exos with HCECs, Exos successfully entered HCECs, indicating their simple endocytosis or internalization by HCECs (Fig. 4(f)). Moreover, the organic influence of MSC-Exos integrated OGG/CMCS hydrogel on HCECs was investigated utilizing a wound therapeutic assay (Fig. 4(g)-4(h)). We detected a major enhancement in scratch closure at 12 and 24 h with the applying of MSC-Exos-loaded hydrogel in comparison with the management (p < 0.001). These outcomes counsel that MSC-Exos maintains long-term exercise for the promotion of corneal epithelium migration in our hydrogel.
The discharge profile of exosomes from the OGG/CMCS hydrogel was proven in Fig. 4(i), indicating a sustained and slower launch of OGG/CMCS-2% hydrogel than OGG/CMCS-1.5% and OGG/CMCS-1% for 72 h after encapsulation of exosomes.
Biocompatibility of OGG/CMCS hydrogels and organic exercise of mesenchymal stem cell exosomes (MSC-Exos) within the hydrogels. (a-b) Cell viability of human corneal epithelial cells (HCECs, a) and keratocytes (HKs, b) cultured with totally different concentrations of hydrogel patch extract following 1, 2, and three days (*p < 0.05 vs. management group; #p < 0.05 vs. Day 1 throughout the identical remedy group). (c) Dimension distribution profile of exosomes (following 1000-fold dilution). (d) Transmission electron microscopy of MSC-Exos. Scale bar: 200 nm. (e) Western blot evaluation of the exosome floor markers. (f) Confocal microscopy of mobile uptake of DiD-labeled MSC-Exos by HCECs after culturing for two h and 12 h. Scale bar: 20 μm. (g) Scratch wound assay. Consultant micrographs of HCECs handled with MSC-Exos-loaded OGG/CMCS hydrogel for 0, 12 h, and 24 h. Scale bar: 100 μm. (h) Quantitative analysis of the scratch wound utilizing ImageJ (**p < 0.01 vs. 12 h throughout the identical remedy group; ###p < 0.001 vs. management group). (i) Launch profile of exosomes from the OGG/CMCS hydrogel
After in vitro characterization and evaluation of the OGG/CMCS hydrogels, we chosen the hydrogel formulation with 2.0% OGG and 1.5% CMCS to evaluate its efficiency in vivo on account of its superior mechanical power, tissue adhesive properties, slower swelling price, and managed launch of exosomes. We implanted the OGG/CMCS hydrogel subcutaneously in mice for 4 weeks to judge its in vivo biocompatibility. Histological examination confirmed that OGG/CMCS hydrogel implantation precipitated no lesions within the coronary heart, liver, spleen, lung, or kidney compared to mice within the management group (Determine S7).
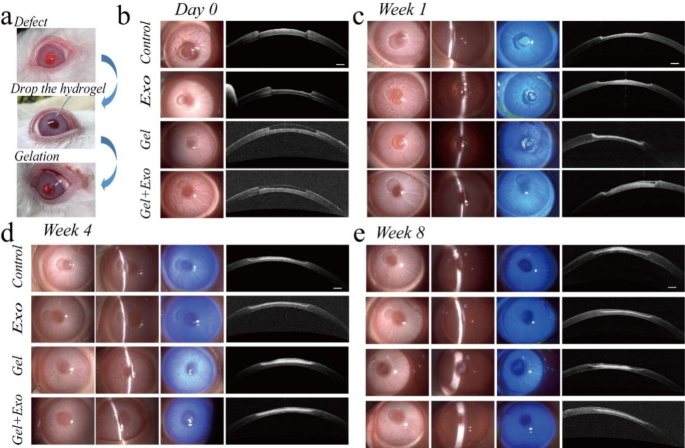
A rabbit corneal defect mannequin was then used to judge the remedy results. The quantity of exosome encapsulation within the hydrogel is 8.75 × 109 particles/mL. The corneal defect was handled with the MSC-Exos-loaded OGG/CMCS hydrogel, pure hydrogel, and MSC-Exos respectively, whereas an untreated group was used because the management (Fig. 5(a)). After gelation, each hydrogels with and with out MSC-Exos adhered successfully to the defected space, exhibiting {smooth} surfaces (Fig. 5(b)). The defected space was monitored in 4 teams at 1, 2, 4, and eight weeks (Figs. 5(c)-5(e), Determine S8). Corneal transparency and the integrity of the corneal floor have been assessed utilizing diffuse illumination and a slim slit beam. The corneal defect space remained {smooth} and clear in eyes handled with MSC-Exos-loaded OGG/CMCS hydrogel throughout the 8 weeks post-surgery, whereas it exhibited gentle opacity within the management and pure hydrogel teams on the eighth week (Figs. 5(c)-5(e)). Additional, fluorescein staining was utilized to look at the corneal re-epithelialization. On the seventh day after surgical procedure, eyes handled with MSC-Exos-loaded OGG/CMCS hydrogel exhibited minimal detection of inexperienced fluorescent staining within the epithelial defect space, contrasting with the comparatively bigger staining noticed within the different teams (Fig. 5(c)), suggesting the novel hydrogel’s potential to reinforce corneal epithelium therapeutic. Anterior section OCT was used to judge the method of hydrogel degradation and wound therapeutic. The hydrogel displayed various mild reflectivity in comparison with the corneal stroma in OCT. The therapeutic course of occurred because the hydrogel step by step degraded and the therapeutic was notably accelerated within the eyes handled with the MSC-Exos-loaded OGG/CMCS hydrogel in comparison with the opposite teams (Figs. 5(c)-5(e) and Determine S8). The hydrogel ultimately degraded fully, leaving no everlasting materials within the cornea.
In vivo evaluation of the MSC-Exos-loaded OGG/CMCS hydrogels in a rabbit corneal defect mannequin. (a) Operation steps. (b)-(e) Consultant slit lamp photographs (diffuse illumination, slim slit beam, and sodium fluorescein staining) and optical coherence tomography (OCT) pictures instantly (b), 1 (c), 4 (d), and eight (e) weeks after surgical procedure. Scale bar: 500 μm
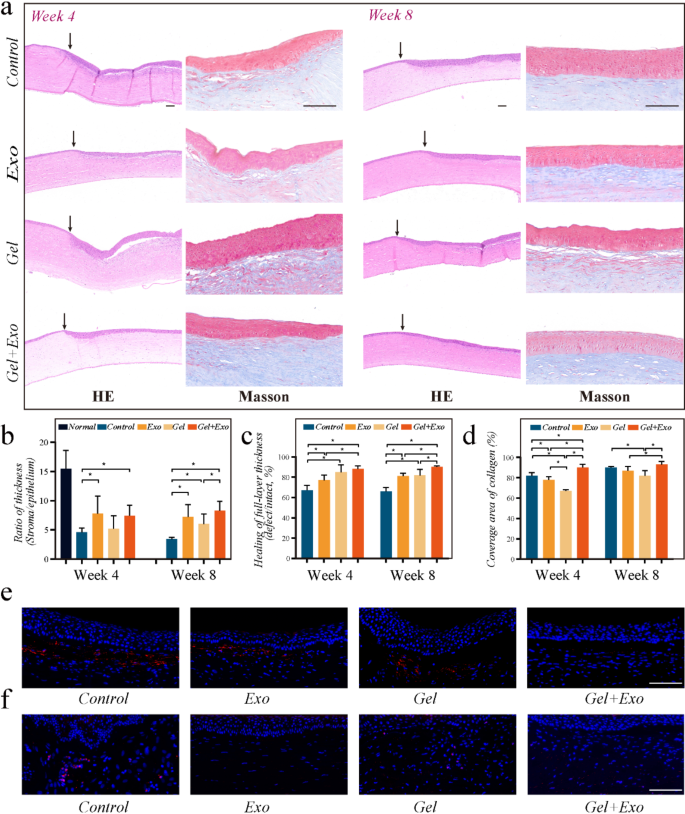
In addition to, histological examinations at 4 and eight weeks revealed corneal restore within the defect space (Fig. 6(a)). In eyes handled with the MSC-Exos-loaded OGG/CMCS hydrogel, the thickness of epithelium and stroma within the defect space carefully resembled that of the encompassing cornea, and the ratio of corneal stroma thickness to epithelial thickness was considerably better than that in eyes with out remedy and eyes handled with pure OGG/CMCS hydrogel on week 8 (Fig. 6(b)). In the meantime, the restoration price of total corneal thickness on week 8 was highest in eyes handled with Exos-loaded hydrogel (Fig. 6(c)). In distinction, the management and pure hydrogel teams exhibited epithelial overgrowth, but the stromal layer and the general corneal thickness have been considerably smaller in comparison with regular circumstances. The exact association of collagen fibrils in stroma is essential for sustaining corneal readability and optimum stromal hydration [1]. Masson trichrome staining illustrated that within the eyes handled with MSC-Exos-loaded OGG/CMCS hydrogel, the stroma maintained a well-arranged lamellar construction in distinction to the eyes handled with pure hydrogel (Fig. 6(a)). The cornea handled with Exos-loaded OGG/CMCS hydrogel exhibited increased protection of collagen fiber (blue) within the corneal stroma in comparison with the Exos and pure hydrogel teams (Fig. 6(d)), indicating its potential to advertise collagen fiber regeneration, whereas vital fibrosis (pink) was noticed within the repaired corneal stroma of Exos and pure hydrogel teams. It could contribute to raised corneal transparency following therapeutic with the help of the exosomes-loaded hydrogel.
Moreover, immunofluorescence staining for alpha-smooth muscle actin (α-SMA) and CD45 was utilized to judge the native fibrosis and inflammatory response in week 4. (Figs. 6(e) and 6(f)). The untreated wounded cornea exhibited increased expression of α-SMA on the wound edge in comparison with the cornea handled with MSC-Exos-loaded OGG/CMCS hydrogels (Fig. 6(e)). Activated keratocytes close to the wound floor seemingly underwent differentiation into α-SMA-positive myofibroblasts within the injured cornea [25]. Exosome-loaded hydrogels considerably decreased α-SMA expression, demonstrating their potential to stop myofibroblast formation and corneal scarring. Leukocyte infiltration, as indicated by CD45 expression, was decrease within the cornea defects handled with exosome-loaded OGG/CMCS hydrogels in distinction to the management and pure hydrogel teams, suggesting the power of exosome-loaded hydrogels to mitigate irritation throughout corneal regeneration (Fig. 6(f)). The findings indicated that the MSC-Exos-loaded OGG/CMCS hydrogel can present an excellent surroundings to reinforce corneal wound therapeutic in vivo.
Histological analysis of operated corneas. (a) HE and Masson staining on week 4 and eight (arrows point out the boundary of the corneal defect). Scale bar: 100 μm. (b) Ratio of corneal stroma thickness to epithelial thickness. (c) Restoration of corneal thickness. (d) Protection space of collagen in corneal stroma. (e-f) Immunofluorescent staining of α-SMA (e) and CD45 (f) on week 4. Scale bar: 100 μm. (*p < 0.05)